
Two parameters describe the helix about this axis: n - the number of residues per helical turn r - the rise per helical residue.Helices are classified as repetitive secondary structure since their backbone phi and psi angles repeat.This means that the dihedral angle pairs phi and psi (phii, psii) are the same for each residue in the helical conformation. In a helical conformation, the relationship of one peptide unit to the next is the same for all alpha-carbons.They are formed when a number of consecutive residues have the same phi and psi angles. A common element of most secondary structures is the presence of characteristic hydrogen bonds e.g., C=O of residue i to HN of residue i+4 (i, i+4).This designation is unfortunate as no portion of a protein’s three dimensional structure is truly random and it is usually not a coil. That which cannot be classified as one of the standard three classes is usually grouped into a category called "other" or "random coil".There are three common secondary structures in proteins, namely alpha helices, beta sheets, and turns.The sausage-shaped regions represent helices, which are arranged in a seemingly irregular manner to form a compact globular molecule. Kendrew's model of the low-resolution structure of myoglobin shown in three different views. 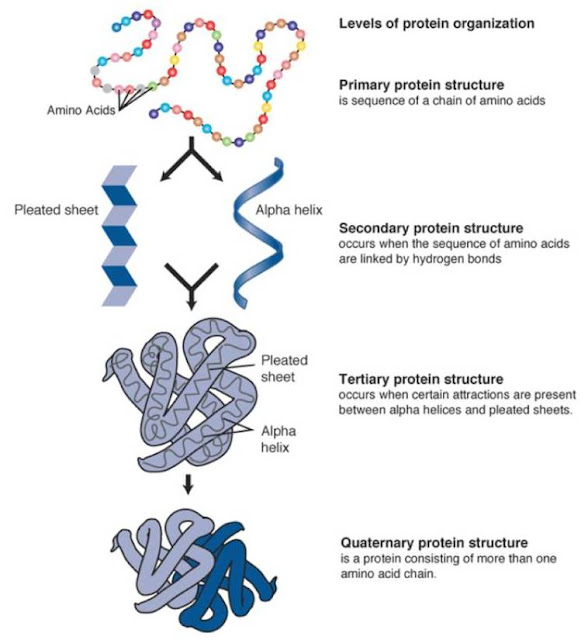
Their Diverse Functions Require Proteins to Have Irregular Structures Secondary StructureMotifs of Proteins Chapter 2
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
